Is that Mpox in Your Balls or Are You Just Happy to See This Preprint?
Deez disordered seminiferous tubules
Note from Angie: Please take my reader survey!! It will only take a few minutes and I want to hear what you think.
Executive Summary
My new year’s resolution is to write about my own research more often. I can’t complain about virologists not doing enough to celebrate virology if I’m not even doing that with my own papers!! So first up: new preprint on a collaboration with Alyson Kelvin to look at whether mpox virus grows in mouse testes or not.
I don’t know why I can’t seem to stay away from the topic of sperm or semen, but this is actually relevant to my research interests. Thankfully, RFK Jr is not depositing papers about teen fertility in my Twitter replies about it. He should be more concerned with the semen of adult men who get mpox, since that is a known threat to public health! Here’s a summary1 of what we found:
What’s The Problem: Mpox is transmitted by sex, but we don’t know if it is transmitted by semen because we don’t even really have a way to study whether it grows in testes or not
What We Did: My colleague and friend Alyson Kelvin asked me to work on her project infecting mice with MPXV clade IIb to see if it grew in their balls or caused disease
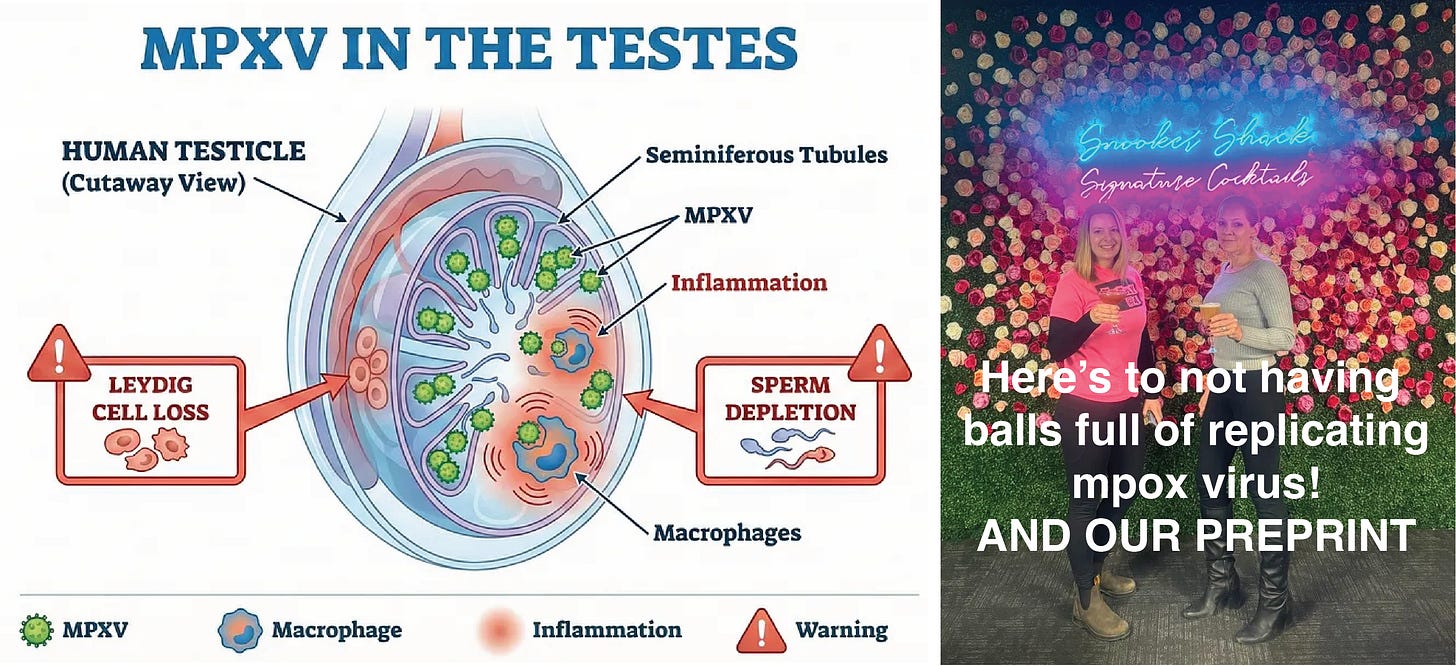
What We Found: MPXV replicates and produces high titres of infectious virus in the testes, possibly by infecting macrophages. This causes observable pathology, including disordered tissues, loss of sperm production, and damage to testosterone-producing Leydig cells.
Public Health Implications: Because the virus replicates in the reproductive tract, sexual transmission may be driven by infectious semen, not just skin-to-skin contact. This raises the risk of presymptomatic transmission before lesions even appear.
Why This Matters: AI can’t predict this level of biological complexity. We need animal models like this to understand how MPXV spreads across tissues and between hosts—especially as global health infrastructure is being dismantled, leaving vulnerable populations at higher risk.
New preprint on mpox in the family jewels
Science news these days is often terribly depressing, so one way I am coping professionally is by continuing to do virology research that is scientifically important and makes an impact on public health. My lab focuses on how the host response to infection impacts pathogenesis, or the process by which viruses cause disease. I focus on emerging zoonotic viruses that pose significant pandemic, epidemic, or security threats. One of these viruses is mpox (monkeypox) virus (MPXV)2, an orthopoxvirus which causes mpox disease. A current burning question in the field is whether or not it infects dudes in the balls.
Mpox causes fever, respiratory illness, and a vesicular rash (pus-filled blisters) that can be localized to the genitals or mouth, or can spread to the entire body. It is painful and can take weeks to resolve. Mpox can be lethal, especially in children and in people who are immunocompromised. It is transmitted by multiple routes, both zoonotic (from animals to humans) and human-to-human. We know sex can drive outbreaks, but we don’t actually know if it is transmitted in semen, because we don’t know if MPXV even productively infects the testes.
I am delighted to announce some ball-breaking news: thanks to Alyson Kelvin at the University of Calgary and an amazing team of collaborators, we have a new preprint out that answers this and provides a new model to follow up on the findings. Alyson and I both use experimental models to study how the host response impacts pathogenesis, although she focuses her work on immune responses and I am more promiscuous in my taste for host responses (which include sex- and sex hormone-related responses). She has contributed extraordinary work to our field for the past 25 years, so I was both privileged and flattered that she invited me to collaborate on this important study. Before she moved to the greener pastures of Calgary, we were office neighbours and frequent cocktail hour companions. She also turned me on to a wandering snack vendor called The Nut Man, who still stocks my office with “Bavarian beer nuts,” pretzels, and popcorn on a biweekly basis. Our friendship was formed over nuts, negronis, and nasty viruses, so this is a perfect collaborative project for us.
I won’t keep you in suspense: YES, this study shows that MPXV infects and fully replicates in testes (and bladder, and the odd penis here and there) in this model and actually causes some observable pathology. Sorry, guys!
Now that the study is out, let’s talk about what we did and why it matters. These findings have a big impact on virology, vaccine development, outbreak containment, and public health. Time to go balls deep into testicular MPXV replication and pathogenesis!
When is sexual transmission really sexual transmission?
Mpox outbreaks have exploded in the past few years, with entirely new evolutionary lineages emerging. Part of the reason these outbreaks have been so large and involved so much transmission is that MPXV spreads through sexual contact. In 2022, a newly emergent clade of MPXV (clade IIb) spread around the world, primarily through sexual networks of men who have sex with men (MSM) and their partners. In 2024, a different newly emergent clade (clade Ib), as well as the former clade I (clade Ia) caused massive outbreaks in the Democratic Republic of Congo, which then spread to other countries. Some continued through 2025. Sexual transmission in a variety of networks, including MSM and sex workers, likely plays a significant role in amplifying and sustaining them.
However, MPXV can be transmitted in many ways, including aerosols and direct mucosal or parenteral exposure to the virus. Is it really sexually transmitted, as in there is virus in semen that infects a new host as the result of sexual interaction? Or is transmission through sex merely circumstantial to features inherent to the act of having sex, since that involves prolonged (if you’re lucky) close, physical contact, friction on skin, with heavy breathing and lots of exposure to aerosols and mucosal surfaces, saliva, and skin? Respiratory, fomite, and direct contact transmission can spread mpox, too. What routes of transmission are actually driving mpox spread during outbreaks, and who is at risk of each?
This is important to know, because if virus is actually replicating in the testes, then that means that it could be transmitted during the prodromal phase of the disease before lesions appear. If this is the case, it creates opportunity for presymptomatic sexual transmission. This has obvious implications for outbreak control, as well as for providing accurate, actionable information and health guidance to the public.
This is also a testable hypothesis. If sexual transmission occurs through virus in semen, blood, or other bodily fluids of the reproductive tract, then the virus will replicate in these tissues. For virus to be transmitted in semen, it first must replicate in the testes. So we need to test whether or not MPXV is replicating in the testes, and if so, how much virus is being produced and whether or not it is causing disease.
In people, we know that there is MPXV in the semen…when studies have looked, and not all the time. There is MPXV DNA in patient semen, and it can stick around for weeks, but that does not mean that this is coming from infectious virus. PCR tests are very sensitive and can detect trace amounts of MPXV DNA, but they can’t distinguish a stray genome from one that is in an infected cell or an infectious virion (virus particle). MPXV DNA is not infectious. Detecting viral DNA genomes does not indicate that infectious virus is being shed into semen in copious quantities, either. Even if there is infectious virus, that does not mean that it is present in sufficient quantity to be transmitted. Infectious virus has been found in semen, but not in every case.
We can’t tell if that is due to technical limitations (specifically, the sensitivity of plaque or median tissue culture infectious dose (TCID50) assays used to quantify infectious virus) or the fact that there really isn’t any infectious virus there. With patients, we don’t always have longitudinally collected samples (collected over time) and semen studies often rely heavily on PCR testing. Regardless of if there is or isn’t virus in semen, that doesn’t tell us anything about how much virus is shed in semen over the course of infection or if or when it crosses a threshold that would present a significant transmission risk.
Apart from transmission, there are also vast knowledge gaps in the reproductive pathology of mpox. If MPXV is indeed infecting the testes, what kind of cells is it infecting? Is it causing disease and if so, how long does that last? Does it determine disease severity and is that related to potential transmission risk? Does testicular mpox infection impact reproductive and sexual health or fertility? What is the impact of vaccination or antiviral treatment on disease or transmission? Are there different infection or disease outcomes for people who are immunosuppressed or co-infected with other viruses?
We can’t really study this in people. You can’t really disentangle the virological questions with the human data that we have now. You can’t subject mpox patients to unnecessary, invasive, longitudinal (repeated) testicular biopsies just to get a look at tissue pathology over the course of infection. Semen studies provide limited information and cannot tell you which cells a virus is replicating in or whether or not they are causing subclinical tissue damage. These also are usually not collected during peak disease since I’d imagine that providing a sample would be a harrowing and excruciating experience with mpox lesions all over your dick. But you can study all of these things in an animal model. So that’s what we did.
Hell yeah, pathogenesis models
For this study, we used a mouse strain (CAST/EiJ) that is particularly susceptible to developing disease after MPXV infection. Most lab mice aren’t very susceptible to MPXV infection and don’t develop disease, possibly because they are all inbred lines from the same subspecies of mouse, Mus musculus domesticus. CAST/EiJ mice are inbred, but they are derived from a wild mouse that turned out to be a different subspecies, M.m. castaneus. Fifteen years ago, Bernard Moss, a virologist at the National Institute for Allergy and Infectious Diseases (NIAID), showed that clade I MPXV caused lethal disease in CAST/EiJ mice, which means that you can use them to study mpox pathogenesis.
Experimental Design and Clinical Disease
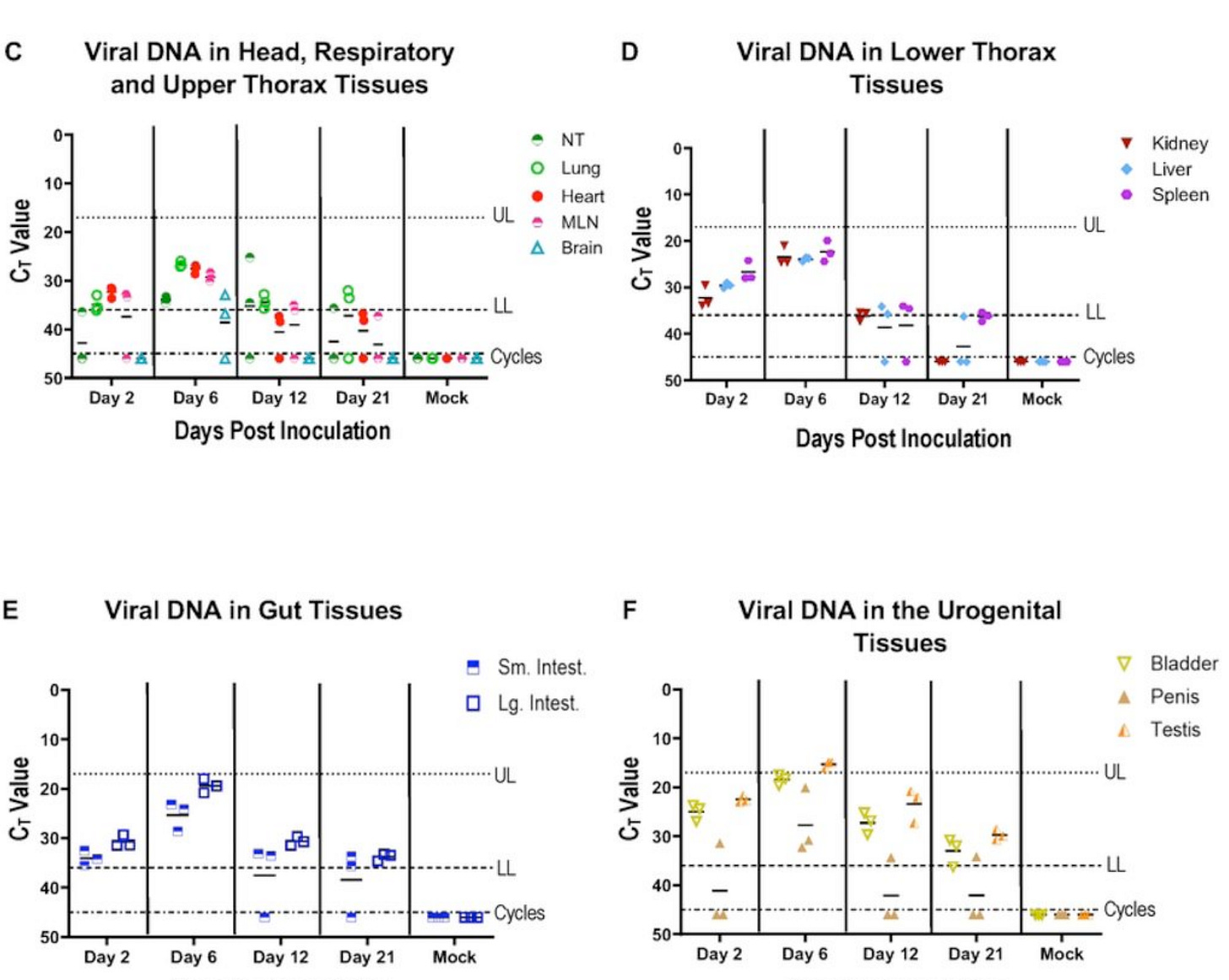
The goal of any pathogenesis model is to mimic a disease condition as accurately as possible, in spite of the limitations that every model has. Sometimes you only mimic aspects of that condition, but that may still be informative, depending on the research question. For MPXV, there are huge knowledge gaps about transmission, and Kelvin conceived this model in part to fill them. It’s a catch-22, because we won’t know if disease is different when mice are infected via different transmission routes, but we can’t study that because we haven’t yet established the model of testicular infection. Kelvin hypothesized that if we infected CAST/EiJ mice by a route that would facilitate spread to multiple tissues, then we would have the best chance of observing whether or not the virus could spread to and replicate in their balls and whether or not it caused disease.
We infected mice intraperitoneally and intranasally with a clade IIb MPXV isolate, which is the virus that caused the 2022 outbreak (and is causing an ongoing epidemic in Africa, although Nigeria has initiated a vaccination campaign to control it). Intranasal infection did not produce any measurable disease and the mice didn’t really develop a productive infection. This is probably strain-dependent, as clade IIb viruses are known to be less pathogenic than clade I. Perhaps CAST/EiJ mice are less susceptible to them by intranasal route.
Intraperitoneal injection is basically injecting virus straight into the main body cavity. The peritoneal cavity contains almost all the digestive organs and a lot of immune organs too, including the spleen, lymph nodes, and specialized immune tissues embedded in tissues of the gut. It’s full of cells called macrophages, which are essential first responders to virus infection. Viruses often LOVE to infect them and/or screw around with their inflammatory cytokine responses, which usually results in more disease. That’s (mostly) what causes Ebola. There is also lots of access to both the circulatory system and lymphatic system, and from there, the rest of the body. And there are neurons connected to the peritoneal organs and abdominal muscles that connect with the central nervous system. That’s how I used to give mice polio back in my PhD student days. The intraperitoneal route is a convenient way to bathe a bunch of different potentially susceptible cells with virus and establish an infection.
Intraperitoneal is also easy to do (much easier than intravenous on a mouse, ask anyone who has ever dealt with tail vein injections), which is critically important when you are doing a high-risk procedure like handling a syringe filled with a highly pathogenic virus like MPXV in containment level 3+ that is tipped with an infectious sharp intended to be stuck into a living vertebrate that can also bite and scratch (CAST/EiJ mice are a fast and somewhat aggressive strain). Easy procedures are often safer procedures, because they create fewer opportunities for something to go wrong that results in a high-risk exposure.
For diseases like mpox, with a broad range of disease severity and extremely complex, multi-variate mechanism of pathogenesis involving multiple cell types and organ systems including the immune system, blood, skin, nerves, and the respiratory and digestive tracts, sometimes just blasting a big bolus of virus right into the peritoneal cavity is the way to go. If the virus normally has to spread across tissues to faithfully replicate human disease—which is the case for mpox infection of the testes—then this is also a good route, since the testes are not in the peritoneal cavity. If we detect virus there, it’s because it spread there. In this case, we don’t need to start by simulating a natural transmission route, because we need to establish whether MPXV can spread to and replicate in the testes. Intraperitoneal injection is a good way to set a baseline for this type of model.
Clade IIb is less pathogenic than clade I viruses. Although it can cause a miserable disease (lesions in very sensitive places, fever, pain) and people can remain symptomatic and contagious for a long period of time, it fortunately doesn’t kill most of the people infected with it (it does, however, kill some people, especially if they have untreated HIV). As such, the mice did not die and there were no differences in body weight over the course of infection. In mice, this means they weren’t sick enough to stop eating. It’s a crude measure, but body weight is one way to assess how sick the mice are getting since they can’t tell us how they feel. These data suggest that these mice probably didn’t feel very sick.
Where is the virus in the body?
Because PCR is sensitive and it is relatively quick to do once the inactivated DNA comes out of containment, it’s a convenient way to detect where the virus went. PCR can only detect the viral genome, so it doesn’t tell us if the virus is replicating or infectious. Viruses get to different places in the body by different mechanisms: free virions (virus particles) in blood or lymph, they can infect cells that travel to and from the blood or migrate through tissues from one organ to another, and they can traverse different tissues themselves by spreading from cell to cell. Dead or non-infectious viruses and viral genomes can move around too, and PCR cannot distinguish between these things. But it’s a good way to quickly survey where the virus goes in any form. And if there’s a lot of virus genome, it indicates that there is probably an infected cell in the vicinity where those genomes are being replicated.
We found MPXV DNA in the nose, lungs, heart, spleen and lymph nodes, kidney, liver, the intestines, bladder, penis, and yes, the testes. Notably, this DNA persisted in the bladder and testes until the end of the experiment at day 21 post-infection. This doesn’t prove that the virus is replicating there, although the Ct values follow a pattern over time that suggests genome replication peaking at day 6 post-infection. So we assessed whether or not we could detect any infectious virus at peak DNA in these same tissues using a plaque assay. The plaque assay detects infectious virus as large dead spots, called plaques, on a layer of cultured cells. You count plaques to calculate infectious titre, or the number of individual infectious viruses in the sample. Infectious viruses are measured in plaque-forming units (PFUs), usually per milliliter of media or milligram of tissue.
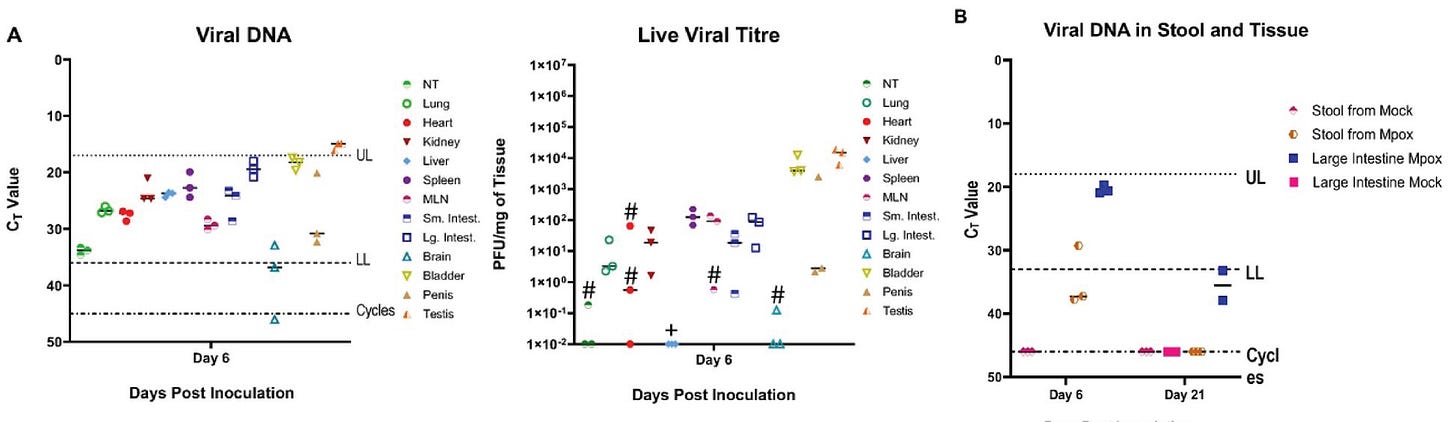
We saw that most of these tissues contained infectious virus, with the intestines, lung, kidney, spleen, and lymph nodes having fairly high infectious titres (more than 100 PFU per milligram of tissue). The highest titres of all were in bladder and testes, although one penis had quite a lot of virus too. These mice had more than 10,000 PFU per milligram of tissue in the testes and bladder. They were firing MPXV on all cylinders; no matter what fluid might be coming out of those mouse penises, it seems like it would probably be full of infectious virus since the cells making up those organs clearly are. These data strongly suggest that cells in both the bladder and the testes are infected and producing high titers of infectious virus.
Is the infection causing any disease at all?
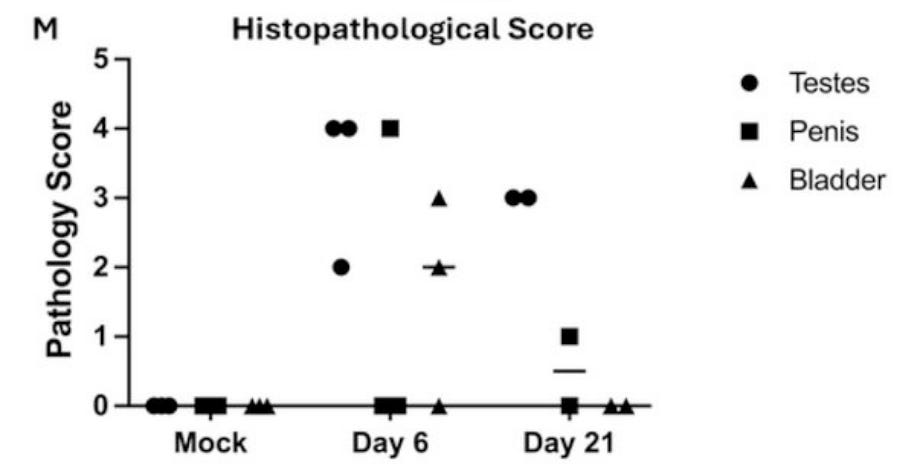
Although these mice didn’t show much morbidity or clinical indicators of sickness, not all damage is visible. Since these mice were very clearly infected, we had a board-certified pathologist rate them for evidence of inflammation using a standardized scoring system. We didn’t see much in most of the other tissues. The testes were another story.
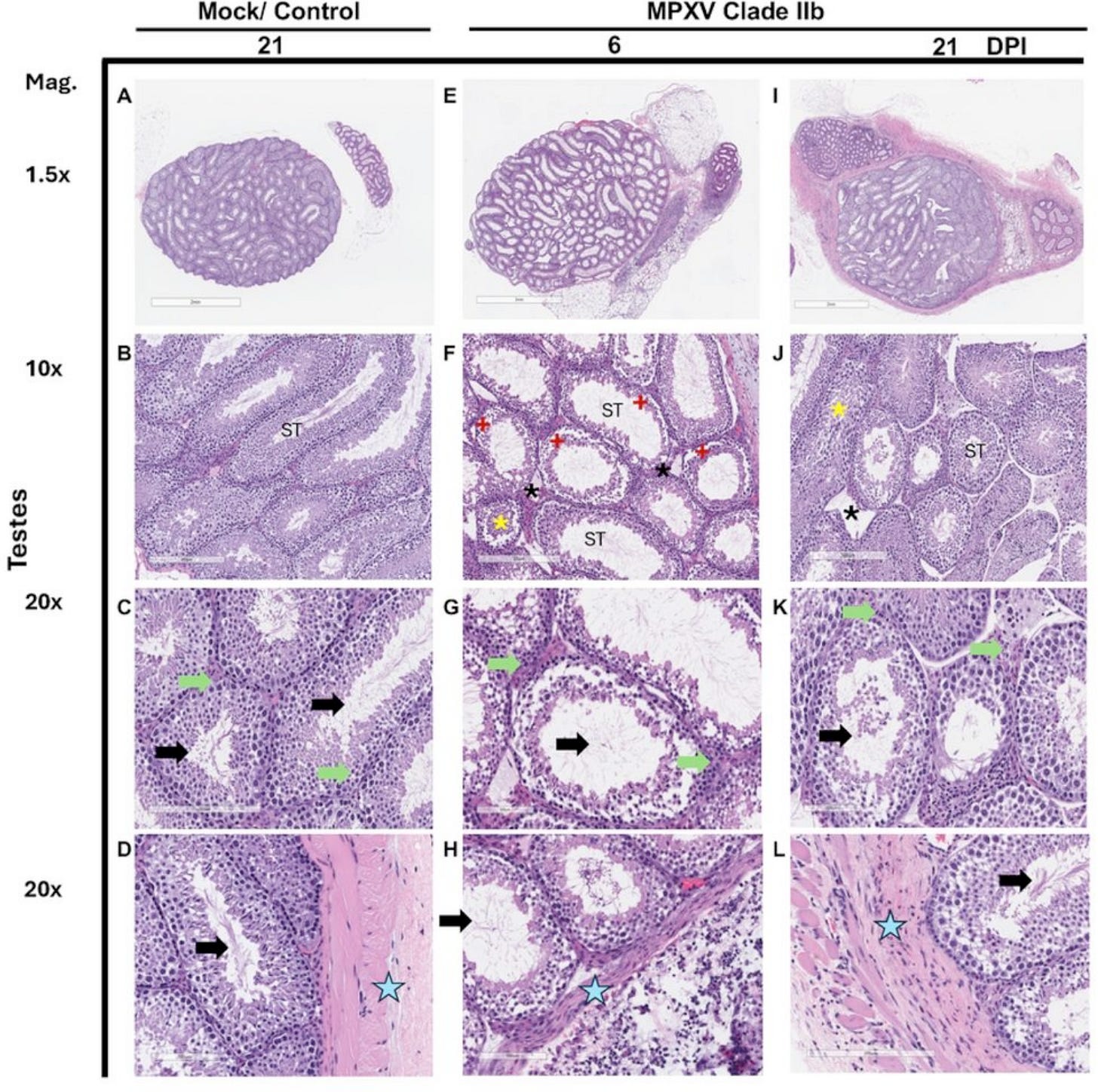
It does not seem to be going very well in the balls of the mice who were infected with MPXV, and things seem especially bad in the seminiferous tubules. These are a bundle of coiled tubes in the testis where sperm develop. Sertoli cells, which support the developing sperm, line these tubules like columns. In the interstitial spaces between the seminiferous tubules, Leydig cells make testosterone. Both of these are required for normal sperm-making testicular function. And both seem to be impacted by the infection. In general, these testes are a mess, with disordered tissue structure, damaged or dying Leydig cells, and loss of sperm production.
The pathology scores measure the observations in the slides according to a standardized scale. These balls are quantifiably not normal. But is it because they are directly infected? Or is it the effect of inflammation caused by the infection in a different tissue? This is what we really want to know: is MPXV replicating and producing infectious virus in cells in the testes?
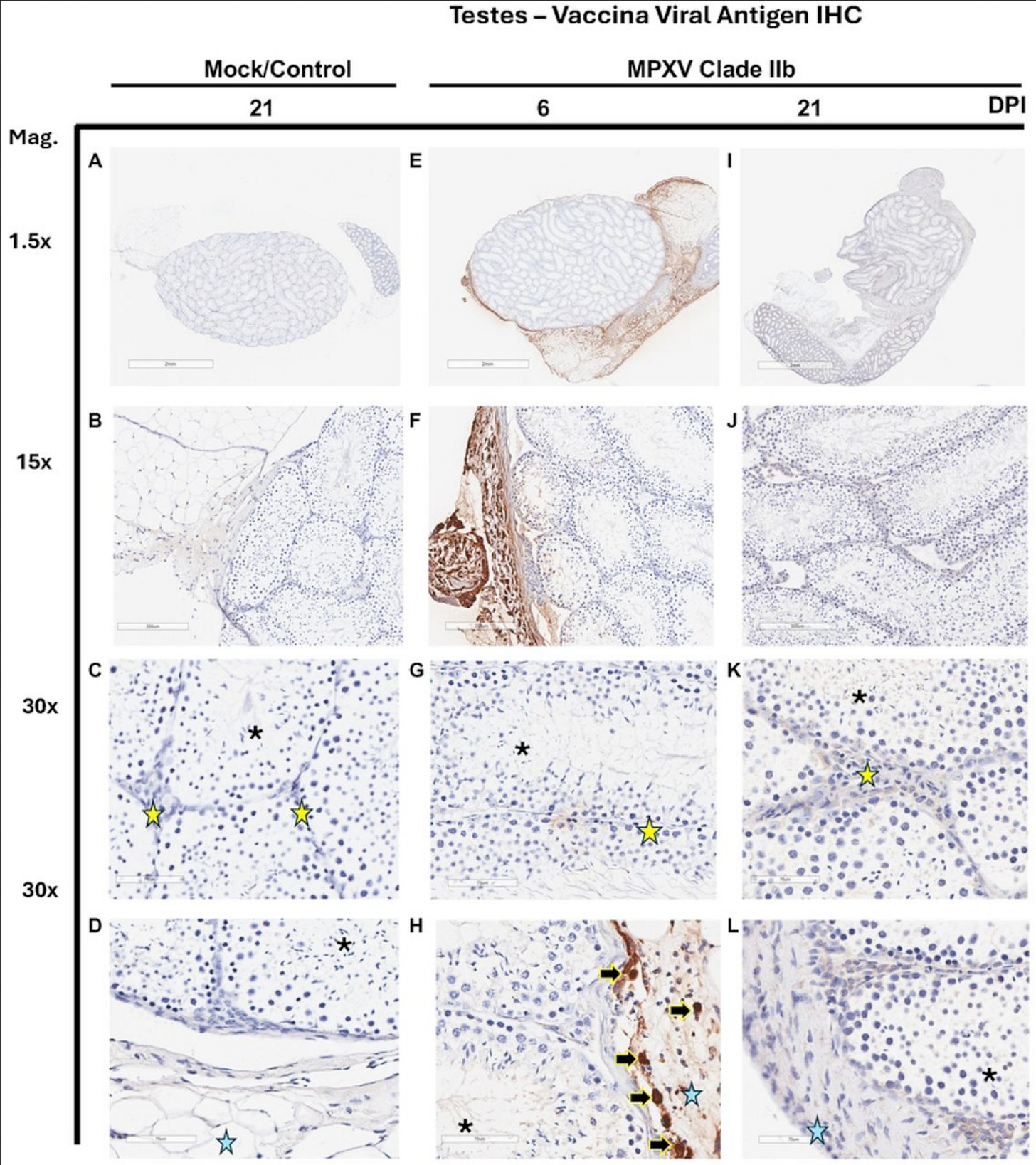
The answer is yes, MPXV does infect the testes! In this stain, we used antibodies against vaccinia virus, another orthopoxvirus that cross-reacts with MPXV (it’s what we use to vaccinate against MPXV) to detect cells producing MPXV proteins. They show up as brown. The middle column shows an awful lot of brown as magnification is increased, while the mock-infected controls show none. These testes are filled with MPXV-infected cells, and they look an awful lot like macrophages.
If they are macrophages, that could explain the impact on the architecture of the entire testicle. Macrophages secrete chemical messengers called cytokines that trigger a variety of damaging inflammatory processes. They also are susceptible to MPXV infection. Viruses that infect macrophages often subvert their mechanisms for regulating this great inflammatory power, which can result in vast tissue damage.
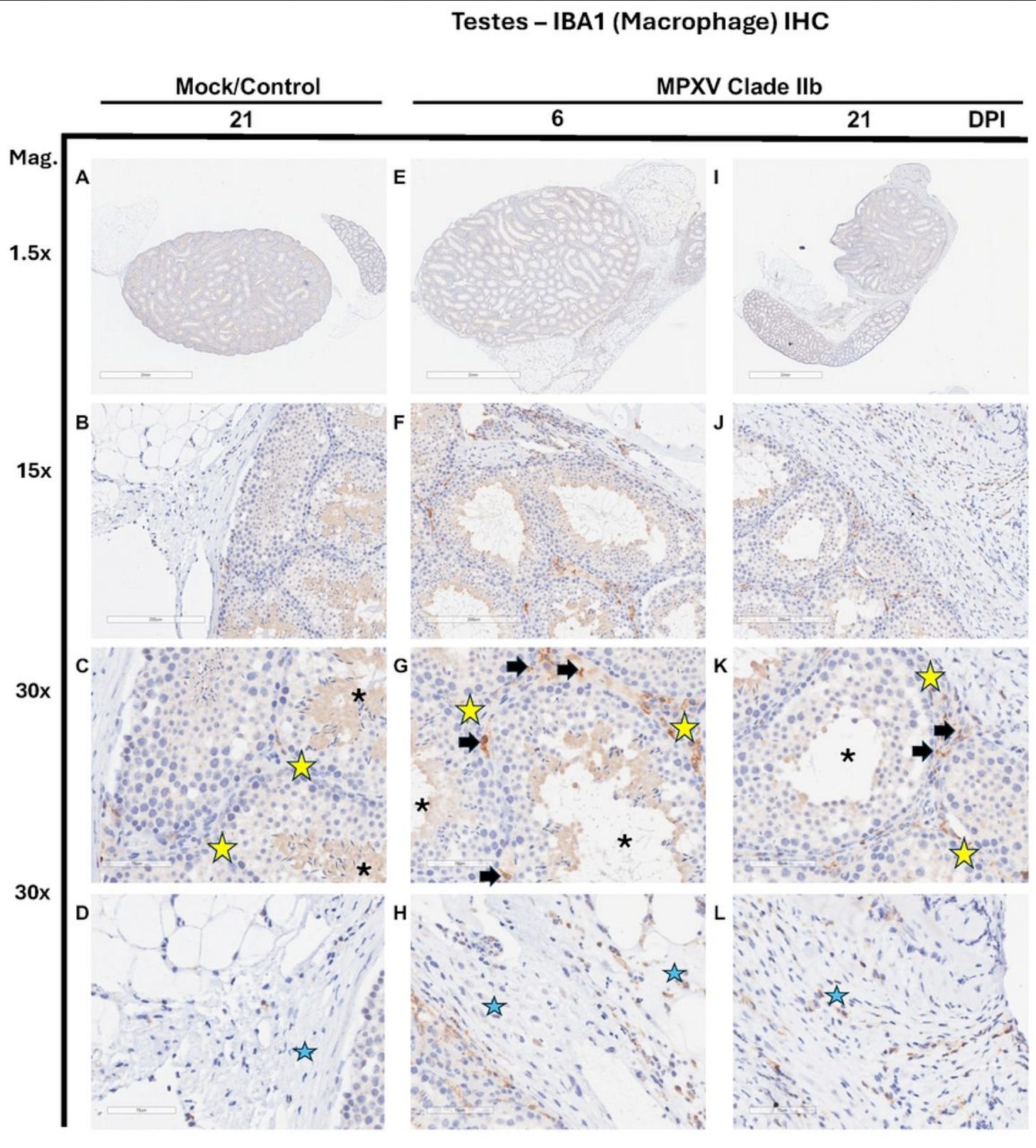
There certainly are plenty of cells that stain brown (for a macrophage marker) in these gonads. They seem to be clustering in the same places that the MPXV-infected cells were (next to the seminiferous tubules and tissue surrounding the testes). This might also explain what is going on: macrophages can have a huge impact on testosterone production by Leydig cells. Testosterone is needed to make sperm and sex hormones can also get up to all sorts of inflammatory mischief, including by signaling to macrophages, that can increase tissue damage. These are examples of hypotheses that can be tested using this model of mpox.
Can we use this model to simulate sexual transmission?
Now that we showed that CAST/EiJ mice can have MPXV infection and pathology in their balls, we wanted to see if they could be infected by routes relevant to sexual transmission. In the global 2022 outbreak, sexual transmission was driven by MSM and their partners, so we started with relevant transmission routes that we could model in male mice: we stuck some virus up their butts and also put some in their mouths.
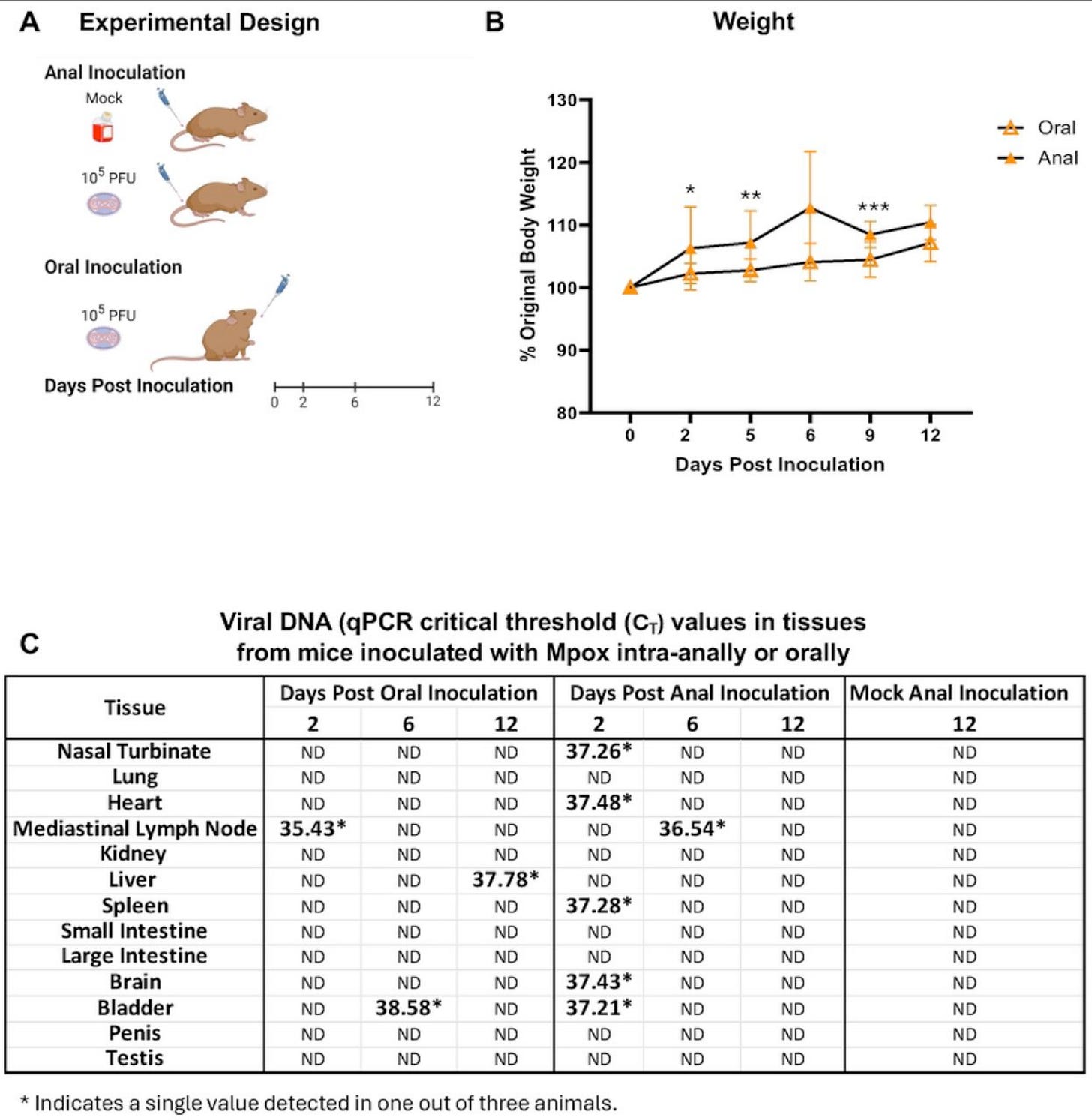
Our hopes of getting some anal or oral infection action were dashed for now. We did detect DNA in some samples, but they were all single animals and Ct numbers were pretty high. They might represent one-off or transient infections. It’s hard to say why this wasn’t a more positive result. Maybe there’s not enough virus present. Maybe establishing an infection requires disruption of the mucosal barrier. Maybe mice aren’t that susceptible by these routes. There are many future avenues to investigate, and thanks to Alyson, we have a model to do it.
Why does MPXV in mouse balls matter?
We already know that sex can drive mpox spread. But because we don’t know exactly whether that is through skin or semen, it is hard to know when an infected person becomes contagious. If transmission is through skin-skin contact, then a person is not likely to be contagious until they develop a rash. If transmission is through semen, then a person might be contagious for days prior to developing symptoms. Presymptomatic transmission makes outbreaks considerably more challenging to contain. Understanding more about the frequency and plausibility of these infection routes is critical to making effective public health decisions and providing accurate guidance. When does a person with mpox present a sexual transmission risk? What is the risk presented by virus in semen versus virus in lesions or breath? Will condoms prevent transmission?
Mpox is popping up more and more frequently. There are a few reasons for this. First, MPXV is zoonotic and circulates in different reservoir species of rodents. As long as people increasingly come into contact with wildlife through climate change, deforestation, habitat loss, resource extraction, and land use and development, zoonotic spillover will continue to occur. Whether or not a spillover turns into a larger epidemic depends on available opportunities to transmit from human to human. There have been a lot of these opportunities over the past few years. There will be a lot more going forward. Violent conflict, political and social upheaval, and remoteness have all hindered efforts to control outbreaks, particularly in DRC. Thanks to the US withdrawing from the World Health Organization, defunding USAID and PEPFAR, and abandoning global health altogether, there’s never been a better time for MPXV to nut up.
Mpox is more likely to cause severe disease if it occurs in someone who is immunocompromised, including people who have untreated HIV. Because of the loss of funding to programs that supported HIV medication for millions of people, there are about to be a lot more people in Africa with untreated HIV, including many in countries with ongoing mpox outbreaks such as DRC. There will be more people who are more likely to have more severe disease.
Mpox can be prevented with vaccination, using the same vaccine that is used for smallpox. For years, there was no momentum to provide a vaccine that until now was primarily made for strategic stockpiles in the event of smallpox bioterrorism, since smallpox was eradicated forty years ago. Recently, vaccination programs have begun in DRC and Nigeria, although huge challenges remain for distribution, regulation, and uptake. In the absence of a vaccination effort that will provide sufficient population immunity, providing guidance that helps people reduce their risk will be essential. That means knowing if mpox can be transmitted in semen or not. To know that, we need to know how MPXV virus gets there, how it grows, and what it does to those cells. With this model, we now have the ability to study that and expand on it.
My lab uses a model called the Collaborative Cross, which is a panel of genetically diverse mice (they are all mice, but genetically different from one another, just like people) to look at how different hosts shape different outcomes when they are all infected with the same dose of the same virus. When we’ve tested these mice with Ebola, high path flu, and SARS-CoV-2, they show a range of disease severity that is entirely dependent on their unique genetic background. In Ebola, some mice die of severe hemorrhagic disease, while others basically lose a little bit of weight and then they are fine. They get “mild” Ebola, because they respond differently to the infection than the ones that die from hemorrhagic disease. It is a great model for trying to understand why some people get very sick and others don’t.
We could potentially use the Collaborative Cross to assess whether or not different individuals have different productive infection and pathology of the testes. These mice are derived in part from CAST/EiJ, so at least some of them will likely have similar infection of the testes. Some of them may have more disease or less. Some may not even have infected testes. Understanding those differences would be tremendously helpful for more accurately assessing individual risks. I would like to expand Alyson’s work into this model, to potentially compare a range of different phenotypes (disease and infection presentations). I am also delighted by the prospect of writing grant applications and academic papers about using machine learning to predict whose balls mpox will ravage.
My lab also is very interested in sex, specifically how sex influences the host response and disease severity. With clade IIb MPXV, there appears to be a male bias toward both cases and disease severity, although this may be simply due to transmission occurring disproportionately among MSM during the 2022 outbreak. Women definitely get infected as a result of sex. I think that it is very important to understand their risk. I’m very interested to see if female CAST/EiJ mice get infected reproductive tissues after intraperitoneal infection. Do they get infected after intravaginal challenge? What risk does mpox present to women as a sexually transmitted infection?
You can be forgiven for thinking that “AIxBio” is anywhere near being capable of replacing animal models of pathogenesis. There is a lot of antipathy, suspicion, and political opposition to animal research, along with a lot of hype for AI (in general, but also specifically in biology). And that’s what it is—hype, because there is no AI model in existence that could have discovered that MPXV clade IIb infects the testes after intraperitoneal infection. They all rely on prior knowledge, and we had none until this study. We cannot replicate the complexity of a virus infecting a mammalian host and spreading across tissues and organ systems, with both direct and indirect impacts on many different cells along the way. But we need models that do contain that complexity to answer these critical questions. Now we have one that can address this for mpox.
Congratulations to Dr. Cynthia Swan (my awesome colleague who led the preprint), Alyson and the Kelvin lab, and all our co-authors. Thanks for inviting me to contribute to this important research.
I’m very long-winded and I know that’s tough for some of these more technical posts. So I’ll put these summaries up on these types of pieces for people who can’t tolerate this much virology even when it’s liberally peppered with dick and ball references.
According to the International Committee on the Taxonomy of Viruses, Orthopoxvirus monkeypox is still called monkeypox virus and not mpox virus. However, this is inaccurate (it was discovered in monkeys but its reservoirs are rodents), stigmatizing for many people in many places for many reasons, and generally unhelpful in every way. For that reason, I’ll just keep using mpox, even when referring to the virus.


Fascinating study. Thank you for contributing to it, and for describing it in this forum.
What is known about mpox replication in the prostate gland or seminal vesicles? Both tissues are important for male fertility & production of ejaculate.
Super sperm is one of those topics that appeals to the manosphere. MPOX was (is?) categorized by many as a disease of "others." So it will be interesting to see if MPOX hitting so close to deez home bridges the divide. Cool research, thanks for the chuckles, too :)